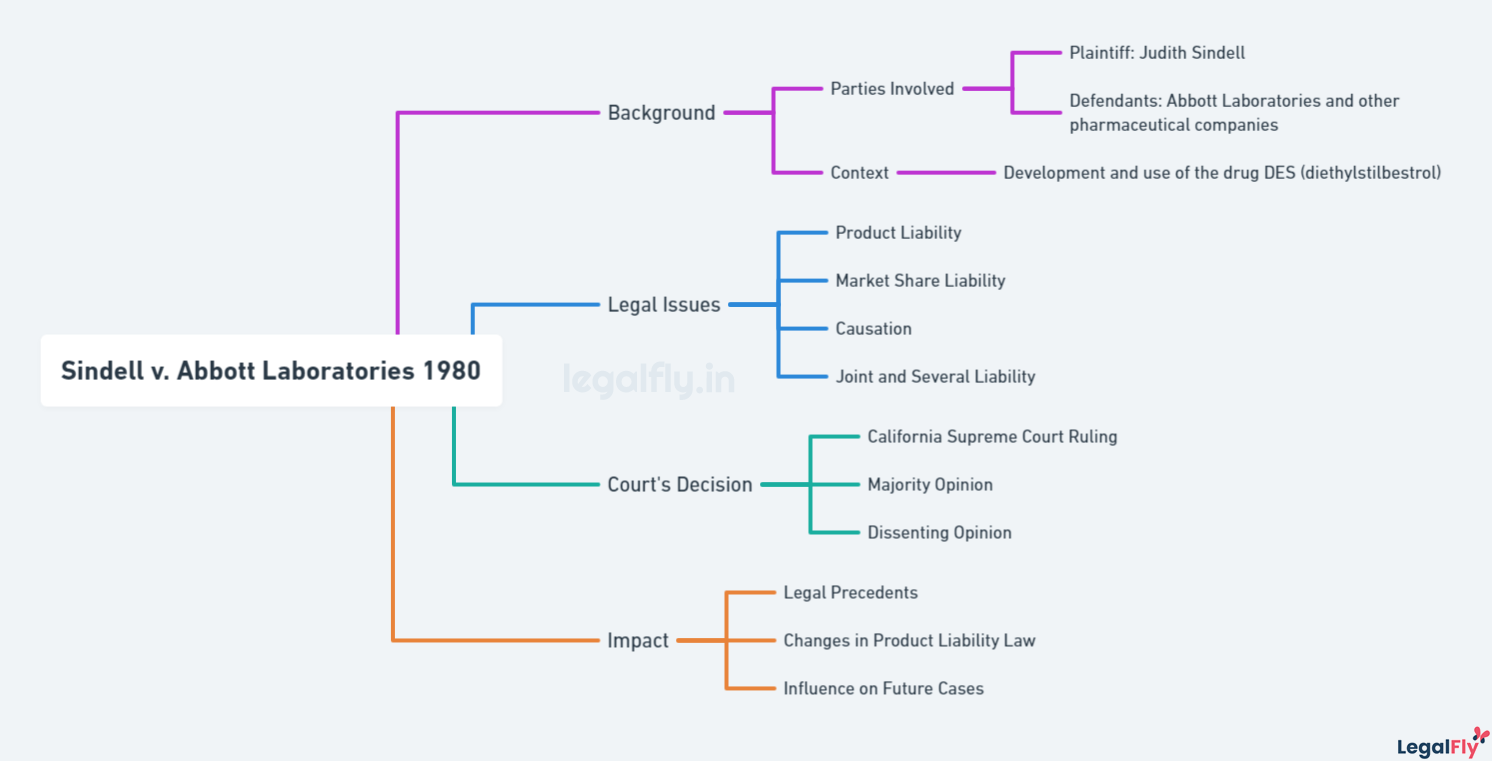
Case Name: Sindell v. Abbott Laboratories
Court: Supreme Court of California
Year: 1980
Citation: 26 Cal.3d 588, 607 P.2d 924, 163 Cal.Rptr. 132
Table of Contents
Introduction to Sindell v. Abbott Laboratories
Sindell v. Abbott Laboratories was a landmark 1980 California Supreme Court case that established the doctrine of market share liability for personal injury cases. This allowed plaintiffs to hold multiple defendants liable for injuries even when the specific manufacturer of the product that caused harm could not be identified.
The case involved women who developed cancer after their mothers had taken the drug Diethylstilbestrol (DES) while pregnant. The plaintiffs could not identify the specific manufacturers of the DES taken by their mothers. The California Supreme Court ruled that the defendants who produced a substantial share of the DES market could be held liable for the injuries based on their market share.
This pioneering doctrine significantly expanded liability for manufacturers and provided recourse for plaintiffs in product liability cases with indeterminate defendants. It has had a lasting influence on tort law and policy. Sindell v. Abbott Laboratories demonstrated the court’s willingness to adapt traditional tort theories to address challenging causation issues arising from modern products and markets.
The Background of the Case
The case involved Judith Sindell, who filed a lawsuit in 1976 against eleven drug companies that manufactured Diethylstilbestrol (DES). DES was a synthetic estrogen prescribed to pregnant women between 1947 and 1971 to prevent miscarriages and other pregnancy complications. Sindell’s mother took DES while pregnant, and Sindell developed a rare vaginal cancer at age 19 in 1971.
Sindell alleged that DES caused her cancer and that the defendant companies were jointly liable even though she could not identify the specific manufacturer of the DES her mother took. At the time, about 200 drug companies manufactured and marketed DES under various names. Identifying a specific manufacturer was difficult because DES was produced from a common industry formula, and patients rarely knew the drug’s brand name.
DES was later shown to be ineffective at preventing miscarriages. Further, it was linked to rare vaginal cancers in daughters exposed in utero. Sindell’s complaint alleged negligence, concert of action, alternative liability, and industry-wide liability among DES manufacturers. She argued the companies failed to adequately test DES before marketing it to pregnant women.
Legal Issues Addressed
The key legal issue addressed in Sindell v. Abbott Laboratories was the challenge of proving causation in cases involving generic products like DES. Hundreds of companies manufactured DES in a generic formulation with no distinguishing features. As a result, plaintiffs had difficulty identifying the specific manufacturer responsible for the DES that caused their injuries.
Traditionally, plaintiffs had to prove that a specific defendant’s product caused their harm in order to hold them liable. However, the Sindell court recognized that this created an impossible burden for plaintiffs in DES cases. Requiring identification of the specific manufacturer would allow companies to avoid liability simply because DES was produced generically.
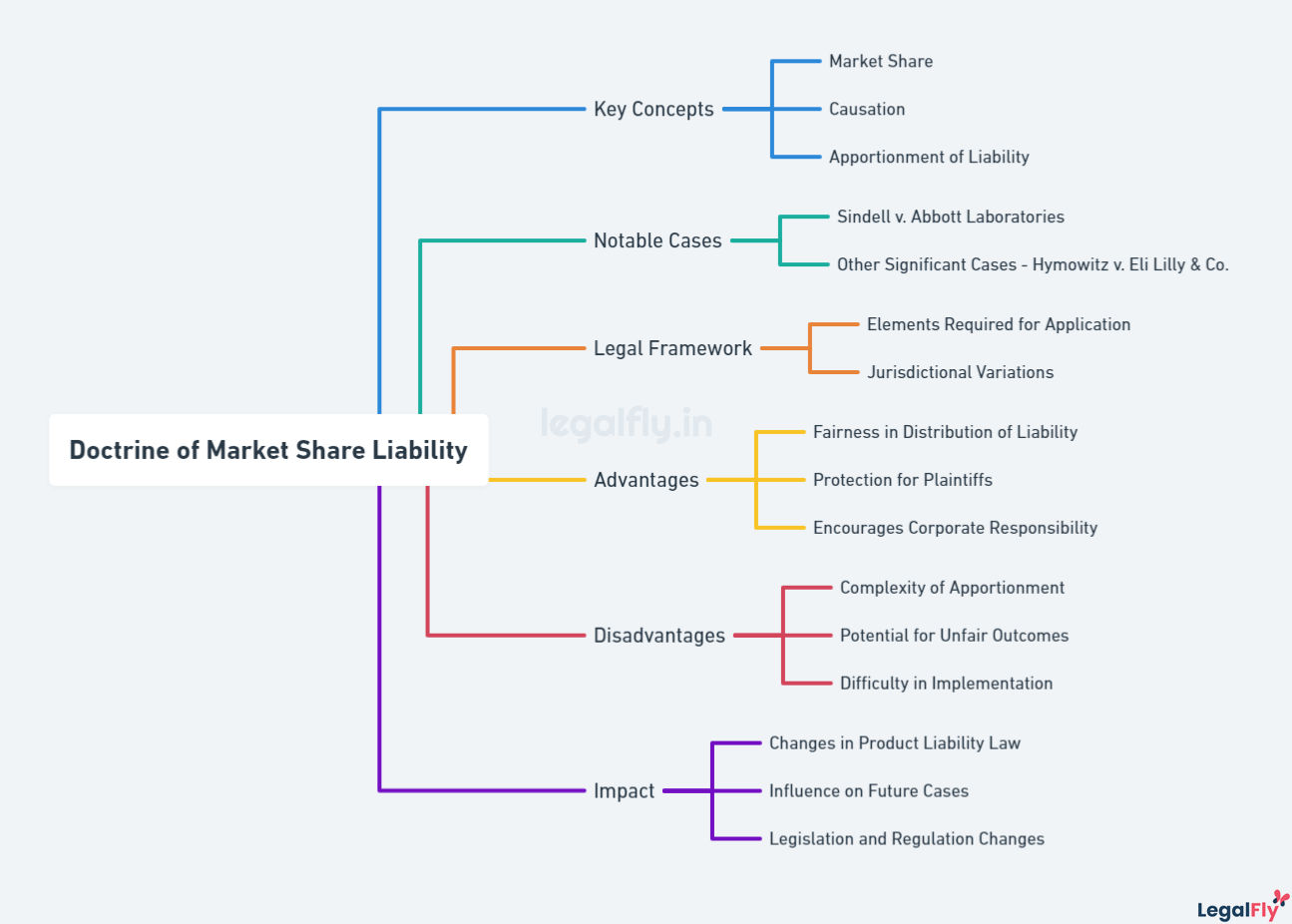
To address this challenge, the California Supreme Court pioneered the doctrine of market share liability. This allowed plaintiffs to hold all DES manufacturers liable based on their respective market share rather than having to single out one company. The court reasoned that liability should be apportioned according to market share since all DES posed the same risk of harm to plaintiffs. This innovative approach enabled plaintiffs to obtain relief despite being unable to pinpoint the exact producer responsible.
The Court’s Analysis
The California Supreme Court’s analysis in Sindell v. Abbott Laboratories centered on the issue of causation and how the plaintiffs could prove which defendant caused their injuries when the generic drug DES was manufactured by hundreds of companies. The court acknowledged the immense challenge facing the plaintiffs due to DES’s “fungible” nature and the long latency period of the injuries.
To address this, the court’s majority opinion, written by Justice Mosk, adopted a new doctrine called “market share liability.” Under this, the defendants would be liable based on their share of the DES market unless they could prove they could not have manufactured the product that caused the plaintiff’s injuries. The court reasoned this approach was justified by balancing policy considerations of providing a remedy to injured parties versus fairness to defendants.
The dissent, written by Justice Richardson, argued against this departure from traditional causation standards. He contended it was improper to hold defendants liable who were not proven to have caused the harm and that the new doctrine compromised their due process rights. However, the majority felt the policy aims outweighed these concerns.
Impact of the Decision
Sindell v. Abbott Laboratories significantly impacted product liability law, particularly for cases involving generic products like DES where the specific manufacturer could not be identified.
The creation of market share liability influenced many subsequent product liability cases by providing an avenue for plaintiffs to seek recovery even when traditional causation could not be proven. Plaintiffs in other states began bringing lawsuits against DES manufacturers using the market share theory established in Sindell. Some state courts adopted the doctrine, while others rejected it or applied variations. Over time, most states accepted some form of market share liability.
Since the Sindell decision, market share liability has evolved in its application. Many courts require plaintiffs to make reasonable efforts to identify the specific manufacturer responsible before invoking market share. Some states only allow market share liability for fungible goods like DES that are chemically identical. Others limit it to particular causes of action or require defendants’ conduct to rise above mere negligence. The theory remains controversial, with critics arguing it erodes traditional causation principles.
The pharmaceutical industry also changed practices in response to Sindell and the threat of market share lawsuits. Companies began improving product labelling, recordkeeping, and testing to help identify manufacturers and minimize liability exposure. Some manufacturers stopped making generic DES to avoid being targeted in market share claims. Overall, the case made manufacturers more cautious regarding safety and liability risks.
Critical Reception and Controversy
The legal community’s response to Sindell v. Abbott Laboratories was mixed. Some lauded the decision as an innovative solution to the challenging issue of proving causation in product liability cases. However, others criticized the adoption of market share liability as going too far.
Supporters of the Sindell ruling argued that it allowed plaintiffs to hold companies accountable when identifying the specific manufacturer responsible was impossible. This was seen as a fair compromise that enabled plaintiffs to have their day in court. As one analysis noted, “Sindell apportioned the defendants’ liability for injuries caused by DES according to their share of the market at the time the drug was ingested by plaintiffs’ mothers”. This was viewed as an equitable approach.
However, critics contended that market share liability was an extreme doctrine that unfairly held manufacturers liable based merely on their market share rather than actual culpability. Some argued that it would open the floodgates to litigation and have a chilling effect on innovation. The dissenting judges in Sindell worried it would force companies to become “insurers of their industry”. They argued market share liability deviated too far from traditional tort law principles of causation.
While some praised Sindell as a pragmatic solution, others felt it set a dangerous precedent. The ruling generated significant debate regarding the appropriate balance between consumer protection and legal doctrine. This controversy persists as courts continue to grapple with applying market share liability principles today.
Lasting Legacy of Sindell v. Abbott Laboratories
Sindell v. Abbott Laboratories has had a profound and lasting impact on product liability law. The market share liability doctrine established in this case fundamentally changed how plaintiffs can seek recourse when injured by a generic product made by multiple defendants.
The case remains highly relevant today as issues of proving causation with generic products persist. Sindell’s precedent allows plaintiffs to hold entire industries accountable rather than placing the burden of proof solely on individuals. This has significant implications for manufacturers, incentivizing safer industry practices and requiring defendants to pay damages based on their market share.
Notable cases influenced by Sindell include Hymowitz v. Eli Lilly & Co., which further refined market share liability for DES cases in New York. Other courts have applied the doctrine to asbestos litigation, lead paint lawsuits, and MTBE groundwater contamination cases.
While some jurisdictions have rejected market share liability, Sindell remains good law in California, and its reasoning continues to shape product liability decisions today. By pioneering an innovative approach to causation, Sindell endures as a landmark ruling.
Conclusion
The California Supreme Court’s decision in Sindell v. Abbott Laboratories was groundbreaking in establishing the doctrine of market share liability. This allowed plaintiffs to hold multiple defendants liable for injuries caused by a fungible product, even when the specific manufacturer could not be identified. By shifting the burden of proof to the defendants to show they were not responsible for the harm, the court enabled plaintiffs to overcome the previously insurmountable barriers of proving causation.
The adoption of market share liability fundamentally altered product liability law. Pharmaceutical companies and other manufacturers are now incentivized to take greater precautions in producing and marketing their goods, knowing they can be held liable based on their market share. Plaintiffs have an avenue to receive compensation for injuries that may have otherwise gone unredressed. While controversial at the time, market share liability has gained broader acceptance in the decades since Sindell. Most states recognize some form of the doctrine today.
Moving forward, it is likely that market share liability will continue to develop as courts adapt tort law to better address the complexities of mass production and marketing. As new industries, such as artificial intelligence, emerge, the reasoning behind the Sindell case may also be applied to hold technology companies accountable. The legacy of balancing individual rights, corporate responsibility, and public welfare will continue to be the lasting impact of Sindell.




